When it comes to brain tumors, the terms glioma and glioblastoma are often used interchangeably, even though they describe conditions with very different behavior and outcomes. Both tumors arise from glial cells, which support and protect nerve cells in the brain. However, glioblastoma represents the most aggressive and life-threatening form within the broader group of gliomas, and its diagnosis carries very different treatment and prognosis implications.
Understanding the distinction between glioma and glioblastoma is critical for accurate diagnosis, treatment planning, prognosis assessment, and patient counseling. The type and grade of the tumor influence how fast it grows, how it is treated, and what outcomes patients and families can realistically expect.
This blog explains the difference between glioma and glioblastoma in a clear, structured, and evidence-based manner. It draws on clinical insights, survival data, and treatment guidelines from leading cancer centers.
Glioma vs. Glioblastoma: Exploring the Basics
What is a Glioma?
Brain tumors are broadly classified based on the type of cells from which they originate, as this determines how the tumor behaves and how it is treated. Among these categories, gliomas form one of the largest and most commonly diagnosed groups of primary brain tumors in adults. They develop within the brain or spinal cord and account for a significant proportion of central nervous system tumors seen in clinical practice.
A glioma is a broad umbrella term used to describe tumors that arise from glial cells, the supportive cells that help maintain normal brain function. Because glial cells are present throughout the brain and spinal cord, gliomas can occur in many locations and affect a wide range of neurological functions. Importantly, gliomas are not a single disease entity. They represent a diverse group of tumors that differ in their cellular origin, biological behavior, and clinical course.
Gliomas vary widely in how fast they grow, how aggressive they are, and how they respond to treatment. Some gliomas grow slowly and may remain stable for years with minimal symptoms, while others grow rapidly and invade surrounding brain tissue. This wide variation means that treatment approaches and outcomes can differ significantly from one patient to another. Tumor grade, molecular features, and location within the brain all play a critical role in determining prognosis and guiding clinical decision-making.
We Handle the Planning
You Focus on Recovery
From Medical Visa assistance to airport pick-up and local stay, HOSPIDIO manages your entire medical trip.
Send Your Treatment Request
Types of Gliomas
Gliomas are further classified based on the type of glial cells they most closely resemble under microscopic examination. This classification helps doctors understand how the tumor is likely to behave and which treatments may be most effective.
1. Astrocytomas:
Astrocytomas arise from astrocytes, the star-shaped glial cells that support neurons and help regulate the chemical environment of the brain. These tumors are the most common type of glioma and can occur at any age. Astrocytomas range from slow-growing, low-grade tumors to highly aggressive forms. Glioblastoma is a Grade 4 astrocytoma, representing the most severe end of this category.
2. Oligodendrogliomas:
Oligodendrogliomas develop from oligodendrocytes, the glial cells responsible for producing myelin, the protective covering around nerve fibers. These tumors tend to grow more slowly than astrocytomas and are often associated with better treatment response and longer survival.
3. Ependymomas:
Ependymomas originate from ependymal cells, which line the ventricles of the brain and the central canal of the spinal cord. These tumors can affect both children and adults and may interfere with the normal flow of cerebrospinal fluid, leading to increased pressure in the brain. Their behavior varies depending on location and grade.
This cell-based classification provides a foundation for understanding the biological differences between gliomas and highlights why treatment and prognosis can vary widely across patients.
Glioma Grades
According to the World Health Organization (WHO), gliomas are graded based on how abnormal the tumor cells look under a microscope.
| WHO Grades | Tumor Behavior | Key Characters | Clinical Implications |
| Grade 1 | Slow growing | Often well defined, non-invasive, ad close to benign | Frequently treated with surgery alone; long-term survival is common |
| Grade 2 | Low-grade but infiltrative | Slow growing but spreads into nearby brain tissue | Requires long-term monitoring risk of progression to higer grades |
| Grade 3 | Malignant and fast growing | Abnormal cells with rapid division | Needs combined treatment with surgery, radiation, and chemotherapy |
| Grade 4 | Highly malignant | Rapid growth, tissue invasion, and areas of necrosis | Gliobastoma falls exclusively under Grade 4 and requires aggresive treatment |
What Is a Glioblastoma?
Glioblastoma, also known as glioblastoma multiforme, is the most aggressive and lethal primary brain tumor seen in adults. It accounts for a large proportion of malignant brain tumors diagnosed worldwide and is known for its rapid progression and poor prognosis. Unlike many other brain tumors, glioblastoma grows quickly and spreads into surrounding brain tissue, making complete surgical removal extremely difficult. Glioblastoma is not a separate disease category, but rather the highest-grade form within the astrocytoma spectrum.
It is classified as a Grade 4 astrocytic glioma under the WHO brain tumor grading system. This classification reflects its highly malignant nature, marked by uncontrolled cell growth, extensive invasion into nearby brain structures, and areas of tumor cell death due to insufficient blood supply. One of the defining features of glioblastoma is its resistance to treatment. Even with aggressive therapy that includes surgery, radiation, and chemotherapy, the tumor frequently recurs. Its ability to infiltrate normal brain tissue at a microscopic level allows cancer cells to remain behind after treatment, contributing to high recurrence rates. These biological characteristics make glioblastoma one of the most challenging brain tumors to manage in clinical practice and highlight the need for ongoing research into more effective therapies.
Biological Features of Glioblastoma
Glioblastoma is defined by several distinct biological features that explain its aggressive behavior and poor response to treatment.
- Rapid cell divisionGlioblastoma cells divide at a very high rate, leading to fast tumor growth and quick worsening of symptoms. This rapid proliferation allows the tumor to increase in size over a short period of time, often causing sudden neurological decline.
- Areas of necrosisAs the tumor grows faster than its blood supply can support, parts of the tumor become deprived of oxygen and nutrients. This results in areas of necrosis, or dead tissue, which are a hallmark feature seen on imaging and pathology in glioblastoma. Necrosis is a key reason glioblastoma is classified as a Grade 4 tumor.
- Extensive blood vessel formationGlioblastomas stimulate the formation of new blood vessels, a process known as angiogenesis. These abnormal blood vessels supply nutrients to the tumor and support its rapid growth. However, they are often poorly structured, which contributes to swelling and leakage in the surrounding brain tissue.
- Infiltration into surrounding brain tissueUnlike tumors that grow as a single mass, glioblastoma cells spread into nearby healthy brain tissue at a microscopic level. This infiltrative growth pattern makes complete surgical removal nearly impossible and is a major reason for tumor recurrence after treatment.
Together, these features explain why glioblastoma behaves more aggressively than other gliomas and why it requires intensive, multimodal treatment approaches.
Glioma vs Glioblastoma: Key Differences Table
| Feature | Glioma | Glioblastoma |
| Definition | Group of tumor from glial cells | Most aggressive Grade 4 glioma |
| Tumor Grade | Grade 1 to Grade 4 | Always Grade 4 |
| Growth Speed | Slow to fast | Very fast |
| Invasiveness | Maybe localized | Highly infiltration |
| Surgerical Removal | Often possible in lower grades | Rarely complete |
| Recurrence Risk | Depends on grade | Very high |
| Median Survival | Years in low-grade tumors | 12-15 months |
| Standard Treatment | Surgery +/- radiation +/- chemotherapy | Surgery + radiation + chemotherapy |
| Prognosis | Wide rane, often favorable low grades | Poor overall |
Glioma is a broad term that includes several types of brain tumors with varying grades, growth rates, and outcomes. Some gliomas grow slowly and can often be managed effectively, especially in lower grades. In contrast, glioblastoma represents the most aggressive end of the glioma spectrum. It is always a Grade 4 tumor, grows very rapidly, invades surrounding brain tissue, has a high risk of recurrence, and requires intensive, combined treatment. As a result, the prognosis for glioblastoma is significantly poorer compared to most other gliomas.
Glioma vs. Glioblastoma Symptoms: Similar Signs, Different Speed
Symptoms of glioma and glioblastoma vary depending on the tumor’s location in the brain, its size, and how quickly it grows. Because different parts of the brain control different functions, even small tumors can cause noticeable symptoms if they affect critical areas. In general, both conditions share many symptoms, but the speed at which these symptoms develop and worsen often differs.
Common Symptoms in Both Conditions
- Persistent headaches
- Seizures
- Weakness on one side of the body
- Speech or vision changes
- Memory problems
Difference in Symptom Progression
- Glioma: Symptoms often develop gradually and may worsen slowly over months or even years, particularly in low-grade tumors. Patients may initially ignore subtle changes, leading to delayed diagnosis.
- Glioblastoma: Symptoms tend to progress rapidly, sometimes over a few weeks. Sudden neurological decline, severe headaches, or new-onset seizures are common and often prompt urgent medical evaluation.
This rapid symptom progression is a key clinical feature of glioblastoma and frequently leads to earlier diagnosis, even though the disease itself is already advanced at the time it is detected.
Diagnosis: How Doctors Differentiate Between Glioma and Glioblastoma
Distinguishing between a glioma and glioblastoma is a critical step in planning the right treatment and understanding the expected disease course. Doctors follow a structured, step-by-step diagnostic process that combines advanced imaging, tissue examination, and molecular testing. Each step adds an important layer of clarity about how aggressive the tumor is and how it should be treated.
1. Brain Imaging (MRI with Contrast)
The diagnostic journey usually begins with a contrast-enhanced MRI scan, which provides detailed images of the brain and helps doctors assess the tumor’s size, location, and behavior.
- Glioblastoma often appears as an irregular, aggressive-looking mass. It typically shows uneven contrast enhancement, significant surrounding brain swelling (edema), and dark central areas that represent dead or necrotic tissue. These features suggest rapid growth and infiltration into nearby brain tissue.
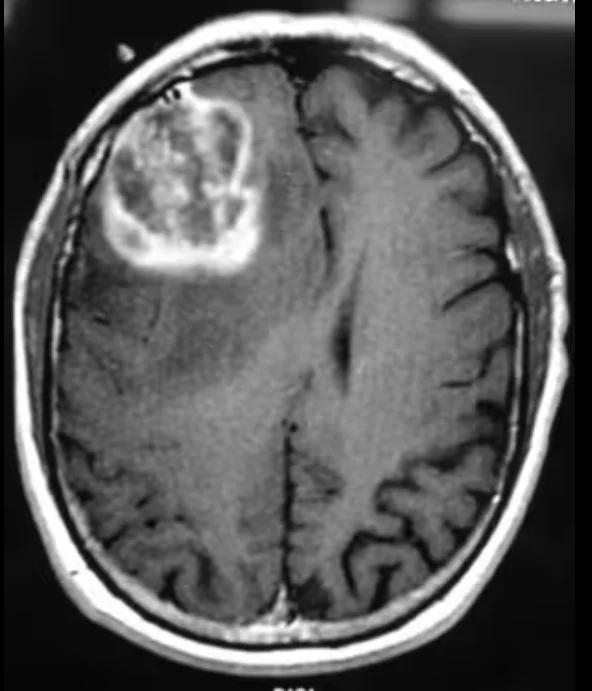
- Lower-grade gliomas, on the other hand, usually look more uniform and well-defined. They tend to grow more slowly, cause less swelling, and often lack areas of tissue death. Some may not enhance strongly with contrast at all.
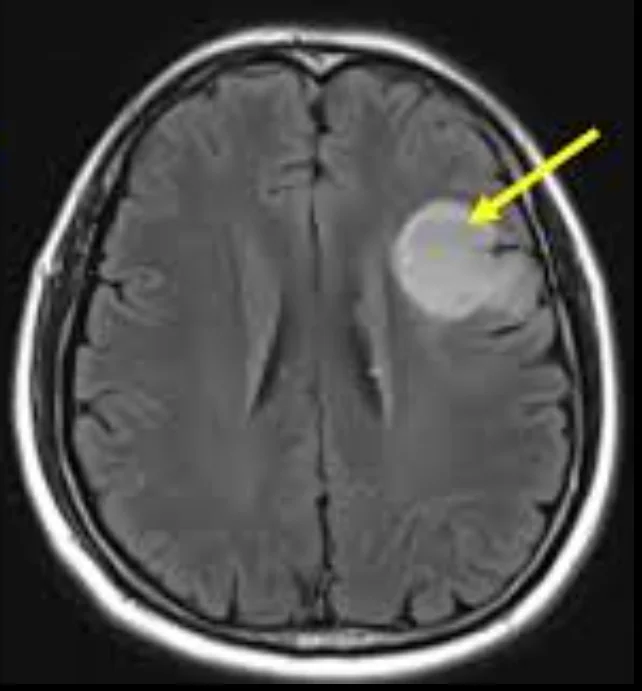
While MRI provides strong clues, imaging alone cannot confirm the exact tumor type or grade.
2. Biopsy or Surgical Tissue Examination
To make a definitive diagnosis, doctors must examine actual tumor tissue.
- This is done either through a stereotactic biopsy (a minimally invasive procedure) or during surgical tumor removal, if surgery is feasible.
- A neuropathologist studies the tissue under a microscope to evaluate cell appearance, growth patterns, and signs of aggressiveness such as rapid cell division or necrosis.
This step is essential to determine whether the tumor is a lower-grade glioma or a Grade 4 glioblastoma, which is the most aggressive form of glioma.
3. Molecular and Genetic Testing
Modern brain tumor diagnosis goes beyond what is visible under the microscope. Doctors now perform molecular testing on the tumor tissue to better classify the disease and personalize treatment.
Key markers include:
- IDH mutation status: Tumors with an IDH mutation generally grow more slowly and are associated with a better prognosis. Glioblastomas are often IDH-wild type, meaning they lack this mutation.
- MGMT promoter methylation: This marker helps predict how well the tumor may respond to chemotherapy, particularly temozolomide. Patients with MGMT-methylated tumors often have better treatment responses.
These molecular features not only confirm the diagnosis but also play a major role in deciding treatment options and estimating outcomes.
We Handle the Planning
You Focus on Recovery
From Medical Visa assistance to airport pick-up and local stay, HOSPIDIO manages your entire medical trip.
Send Your Treatment Request
Why Accurate Diagnosis Matters
Understanding whether a tumor is a glioma or a glioblastoma helps doctors:Choose the most effective treatment planEstimate prognosis more accuratelyAvoid over- or under-treatmentGuide patients and families with clearer expectationsA precise diagnosis ensures that each patient receives care tailored to the tumor’s biology—not just its appearance.
Treatment of Glioma
Treatment for glioma is guided by the tumor grade, molecular profile, size, location, and the patient’s overall health. Because gliomas vary widely in behavior, treatment plans are individualized rather than uniform.
Surgery
Maximal safe surgical removal is usually the first step in treatment whenever possible. The goal is to remove as much of the tumor as can be done safely without damaging critical brain areas.
- Surgery is most effective in low-grade gliomas, where tumors are more localized
- Removing tumor tissue reduces pressure within the brain
- Many patients experience improvement in symptoms such as headaches or weakness after surgery
Radiation Therapy
Radiation therapy is used to destroy remaining tumor cells after surgery or when surgery is not fully possible. Radiation is commonly recommended when:
- The tumor is Grade 3 or higher
- A residual tumor remains after surgery
- The tumor shows signs of progression
Chemotherapy
Chemotherapy is often combined with radiation or used alone depending on tumor type and grade. Common chemotherapy approaches include:
- Temozolomide, widely used for higher-grade gliomas
- PCV chemotherapy (procarbazine, lomustine, and vincristine), particularly effective in certain oligodendrogliomas.
In some cases, low-grade gliomas with favorable features may be closely monitored with regular imaging before starting radiation or chemotherapy, allowing patients to delay treatment-related side effects while maintaining disease control.
Treatment of Glioblastoma
The treatment of glioblastoma is urgent and highly coordinated because this tumor grows rapidly and tends to spread into surrounding brain tissue. Care is managed by a multidisciplinary medical team, typically involving neurosurgeons, radiation oncologists, medical oncologists, neurologists, rehabilitation specialists, and palliative care professionals. Since glioblastoma cannot be cured with a single therapy, treatment relies on a combined, step-wise approach designed to control tumor growth, relieve symptoms, and preserve quality of life for as long as possible.
1. Maximal Safe Surgical Removal
Treatment usually begins with maximal safe surgery, meaning surgeons remove as much of the tumor as possible while protecting critical brain functions such as speech, movement, vision, and memory.
Surgery helps to:
- Reduce pressure inside the brain
- Improve neurological symptoms like headaches, seizures, or weakness
- Provide tissue for accurate diagnosis and molecular testing
- Enhance the effectiveness of follow-up treatments
- Because glioblastoma cells often infiltrate normal brain tissue, complete removal is rarely possible.
Even after extensive surgery, microscopic tumor cells almost always remain.
2. Radiation Therapy with Concurrent Chemotherapy
After surgery, most patients receive radiation therapy combined with chemotherapy, usually starting within a few weeks.
- Radiation therapy targets the tumor bed and surrounding areas where microscopic cancer cells may be present. It is typically given five days a week for about six weeks.
- Chemotherapy (most commonly oral temozolomide) is given alongside radiation to make cancer cells more sensitive to radiation damage.
This combined approach is considered the standard of care and significantly improves survival compared to radiation alone.
3. Maintenance (Adjuvant) Chemotherapy
Once radiation therapy is completed, patients usually continue with maintenance chemotherapy.
- Temozolomide is taken in cycles over several months
- The goal is to slow tumor regrowth and prolong disease control
- Tolerance and response are closely monitored through regular scans and blood tests
The duration of maintenance therapy depends on the patient’s response, overall health, and side-effect profile.
4. Additional and Advanced Treatment Options
In selected patients, additional therapies may be recommended:
- Tumor Treating Fields (TTF): A wearable device that delivers low-intensity electrical fields to disrupt cancer cell division. It may help extend survival when used consistently alongside chemotherapy.
- Clinical trials: Patients may be eligible for trials involving targeted therapy, immunotherapy, vaccines, or novel drug combinations.
These trials offer access to emerging treatments not yet widely available.Eligibility depends on tumor biology, prior treatments, and overall health status.
5. Supportive Care, Rehabilitation, and Symptom Management
Supportive care is a vital part of glioblastoma treatment and continues throughout the disease course.
This may include:
- Medications for seizures, brain swelling, pain, or fatigue
- Physiotherapy, occupational therapy, and speech therapy to regain function
- Nutritional support and psychological counseling
- Palliative care to improve comfort, independence, and emotional well-being
Early integration of supportive care helps patients maintain a better quality of life while undergoing intensive treatment.
Survival Rates in Glioma vs. Glioblastoma: What Studies Show
When it comes to glioblastoma survival, clinical research consistently shows that outcomes remain poor even with current treatments, though there have been modest improvements over time.
According to PubM, Median overall survival for adults with newly diagnosed glioblastoma is typically around 12 to 15 months, even with maximal safe surgery followed by radiation and chemotherapy. This is supported by systematic reviews and large institutional studies showing that the combined standard treatment improves survival compared with radiation alone.
A systematic analysis of glioblastoma outcomes reported a median overall survival of approximately 13.5 months, with five-year survival rates generally below 6%. Another PubMed study reported outcomes ranging from 9 to 14 months median survival, depending on treatment intensity and patient factors.
Survival varies widely based on several key factors:
- Extent of surgical resection: Patients who undergo more complete tumor removal tend to live longer than those with only a biopsy.
- Molecular markers: Tumors with MGMT promoter methylation and IDH mutations are associated with longer survival than tumors without these markers.
- Age and overall health: Younger patients often have better outcomes than older patients.
Even with optimal treatment, long-term survival is rare. Five-year survival rates in contemporary populations are generally low, often in the single digits, reflecting the aggressive biology of glioblastoma. Some older systematic reviews suggest five-year survival of around 4% in many cohorts, and less than 1% may survive ten years or more.
In contrast, lower-grade gliomas (Grades 2 and 3) typically have much longer survival, frequently years to decades in many patients, especially when molecular features and treatment response are favorable. Overall, while median survival for glioblastoma remains around 12 to 15 months, individual outcomes vary greatly based on biological, clinical, and treatment-related factors.
Why Glioblastoma Recurs Frequently?
Despite aggressive therapy that includes surgery, radiation, and chemotherapy, glioblastoma recurrence is common and often unavoidable. Most patients experience tumor regrowth, even when treatment follows the best available standards. One major reason is microscopic tumors spread beyond visible margins. Glioblastoma cells infiltrate surrounding brain tissue at a microscopic level, extending far beyond what can be seen on MRI scans or removed during surgery. As a result, cancer cells are often left behind even after maximal safe resection. Another contributing factor is resistance to chemotherapy.
Glioblastoma cells can adapt and develop resistance to drugs such as temozolomide, reducing the long-term effectiveness of treatment. Certain tumor cells survive initial therapy and later drive tumor regrowth. Limited drug penetration across the blood–brain barrier also plays a significant role. The blood–brain barrier protects the brain from harmful substances but also restricts many chemotherapy drugs from reaching tumor cells in adequate concentrations, limiting treatment impact.
Because of these factors, most glioblastoma recurrences occur within one year of initial treatment. Recurrence is usually more difficult to treat and often requires additional therapy, clinical trials, or supportive care approaches.
Pediatric vs Adult Differences in Glioma vs. Glioblastoma
- Tumor behavior: In children, gliomas are often different from those in adults. Some pediatric gliomas grow more slowly and respond better to treatment, while others can still be aggressive. Adults are more likely to develop glioblastoma, which is a fast-growing, aggressive tumor.
- Treatment tolerance: Children’s bodies and brains are still developing, so they may respond differently to surgery, chemotherapy, or radiation. Doctors carefully balance treatment effectiveness with minimizing long-term side effects on growth and brain development. Adults may face more side effects from aggressive treatments, and recovery may be slower.
- Symptoms and detection: In children, symptoms like changes in school performance, behavior, or coordination may appear first, sometimes before obvious physical signs. Adults often notice headaches, memory changes, or weakness in a limb.
- Long-term considerations: Pediatric patients may need ongoing monitoring for learning, development, and cognitive changes after treatment. Adults may face challenges in returning to work or managing daily responsibilities, and need support for mental health and quality of life.
Quality of Life and Supportive Care
Supportive care for patients with glioma or glioblastoma goes beyond treating the tumor itself; the main goal is to help patients remain as comfortable, independent, and engaged in daily life as possible. Symptom management plays a central role, with medications tailored to control headaches, seizures, nausea, fatigue, and other side effects, while pain management is personalized to each patient’s needs. Rehabilitation services such as physical therapy, occupational therapy, and speech therapy help maintain strength, mobility, daily functioning, and communication abilities.
Emotional and social support is equally important, counseling, support groups, and guidance for families help patients cope with stress, anxiety, or depression. Children may require additional assistance with school, social skills, and emotional development, whereas adults may need help balancing work, household responsibilities, and maintaining independence.
Holistic approaches, including nutrition, gentle exercise, and relaxation techniques, are encouraged to improve overall well-being. Palliative care can also be introduced early to enhance comfort, maintain dignity, and support both patients and their families throughout the treatment journey.
Also Read: Brain Tumor Treatment Cost in India
Clinical Perspective
From a medical standpoint, glioblastoma is not just a more serious form of glioma, it is a different and more aggressive disease with its own behavior and treatment challenges. Unlike lower-grade gliomas, glioblastomas grow faster, affect the brain more severely, and are harder to treat. Early diagnosis using molecular tests (which look at the tumor’s genetic makeup) is very important because it helps doctors choose the most effective treatments for each patient.
Care for these patients usually involves a team of specialists, including neurosurgeons, oncologists, radiation doctors, nurses, and therapists. Working together in this way, doctors can improve symptoms, slow tumor growth, and support quality of life for both glioma and glioblastoma patients.
Conclusion: Glioma vs Glioblastoma
Glioma is a broad category of brain tumors that can behave very differently depending on their type and grade. Some gliomas grow slowly and may respond well to treatment, while others are more aggressive. Glioblastoma, on the other hand, is the most aggressive form of glioma. It grows rapidly, often recurs after treatment, and generally has a more limited survival outlook. Clearly distinguishing between glioma and glioblastoma is critical. It guides doctors in choosing the most appropriate treatments, helps patients and families understand what to expect, and allows caregivers to plan supportive care effectively.
By understanding these differences, patients, families, and healthcare teams can make informed decisions, prepare for challenges, and approach treatment with realistic expectations. This clarity not only supports better medical outcomes but also helps maintain quality of life and emotional well-being throughout the journey.
Read More Related Blogs
References
Recent Blogs

FAQs
A glioma is a broad term for brain tumors that arise from glial cells, which support and protect nerve cells. Gliomas can be slow-growing or fast-growing, and range from low-grade to high-grade tumors.
Glioblastoma (GBM) is the most aggressive and highest-grade (Grade IV) type of glioma. It grows rapidly, infiltrates surrounding brain tissue, and requires intensive treatment.
Yes. Glioblastoma is a type of glioma, but not all gliomas are glioblastomas.
Yes. Some low- or intermediate-grade gliomas can transform into glioblastoma over time, especially if untreated or genetically aggressive.
Currently, glioblastoma is not considered curable, but treatment can: Prolong survival, reduce symptoms and improve quality of life.
Glioblastoma grows rapidly and can progress over weeks to months, which is why prompt treatment is essential.
Survival varies, but with treatment, many patients live 12–18 months or longer, depending on age, health, and tumor biology.
If surgery is not possible, radiation and chemotherapy are used to control tumor growth and manage symptoms.
Markers like IDH mutation and MGMT methylation help guide treatment decisions and predict response to therapy.
Most cases are not inherited. Glioblastoma usually occurs sporadically without a family history.
Sanjana Sharma is a certified diabetes educator with a solid academic background in nutrition and dietetics. Her qualifications include a BSc in Clinical Nutrition and Dietetics, an MSc in Foods and Nutrition from CCS University, a Diploma in Health and Education from IGNOU, and a certification from NDEP. Dedicated to helping patients manage their health through personalized care and education, she brings expertise and compassion to her work. Outside of counseling and writing, Sanjana loves staying updated with fashion trends, sharing corporate memes on Instagram, and, of course, thinking about food.
Dr. Manmohan Singh is a senior neurosurgeon and Vice Chairperson – Neuro Sciences at Paras Health, Gurugram. With over 25 years of experience, he specializes in complex brain and spine surgeries, skull base procedures, and advanced neurosurgical care. He previously served as Professor of Neurosurgery and Head of the Gamma Knife Centre at AIIMS, New Delhi, and is widely respected for his clinical excellence and patient-centric approach.